Ecotropic viral integration site 1 (EVI1) is essential for hematopoiesis, particularly for the maintenance of hematopoietic stem cells. Aberrant expression of EVI1 is widely known to be an independent poor prognostic factor in acute myeloid leukemia (AML). Previous studies have shown that enhancer hijacking or gene fusion caused by chromosomal translocation or inversion involving 3q26 leads to EVI1 overexpression. Intriguingly, however, aberrant EVI1 expression is seen in many cases of AML without 3q26 rearrangement. The prognosis of these cases is similarly dismal, but the mechanism that causes aberrant EVI1 expression in the absence of 3q26 chromosomal abnormalities is largely unknown.
To identify key regulatory factors essential for EVI1 expression in AML, we performed a clustered regularly interspaced short palindromic repeats (CRISPR)/CRISPR-associated protein 9 (Cas9) knockout screen. We knocked in the green fluorescent protein (GFP) coding sequence at the EVI1 locus in the human myeloid leukemia cell line K562, which expresses high levels of EVI1 without a 3q26 chromosomal aberration. After transduction of a genome-wide CRISPR/Cas9 knockout library, transduced cells were separated into GFP High and GFP Low subpopulations by flow cytometry. By sequencing the uniquely attached barcodes, we estimated the effect of gene knockout for each gene. Genes counted more frequently in the GFP Low fraction were listed as candidates for activators of EVI1. The top 13 genes, including three apparent positive controls such as EVI1 and two ribosome-related genes, had a false discovery rate (FDR) of less than 1%. We validated these candidate genes in K562 and two other EVI1-overexpressing AML cell lines by transducing individual single guide RNAs targeting these genes.
These analyses identified zinc finger protein 91 (ZFP91) as a potential regulatory factor that significantly upregulates EVI1 expression common to the three independent myeloid leukemia cell lines. ZFP91 is an atypical E3 ubiquitin ligase, a DNA-binding protein that functions as a transcription factor. ZFP91 promotes cell growth and inhibits apoptosis in AML and has been shown in previous studies to activate multiple oncogenic pathways, including nuclear factor-kappa B (NF-κB), mitogen-activated protein kinase (MAPK), and hypoxia-inducible factor-1α (HIF-1α).
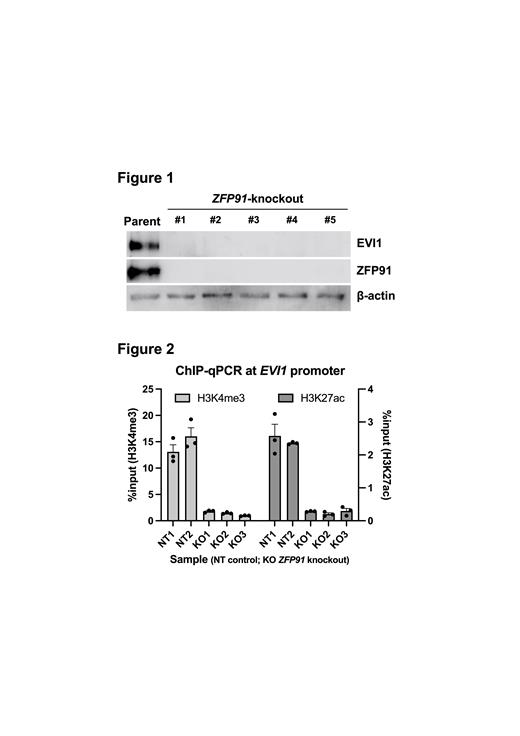
We demonstrated that knockout of ZFP91 in K562 substantially reduced EVI1 protein expression to undetectable levels by Western blotting (Figure 1). EVI1 mRNA expression also decreased dramatically after ZFP91 knockout. These results suggest that ZFP91 may play an indispensable role in EVI1 transcription. We performed chromatin immunoprecipitation quantitative polymerase chain reaction (ChIP-qPCR) targeting the EVI1 promoter in K562 and demonstrated that ZFP91 knockout clones showed approximately 10-fold weaker signals for active histone modifications (H3K4me3 and H3K27ac) at the EVI1 promoter (Figure 2). In addition, chromatin immunoprecipitation sequencing (ChIP-seq) for ZFP91 in K562 revealed the presence of a distinct peak as well as the ZFP91-specific motif sequence within the EVI1 promoter region. These data strongly suggest that ZFP91 functions as an essential transcription factor for EVI1 gene overexpression in some EVI1-associated myeloid leukemias. We confirmed that ZFP91 knockout in K562 inhibited cell proliferation, which is consistent with previous studies in other AML cell lines. Based on these results, we believe that ZFP91 has the potential to serve as a therapeutic target in EVI1-associated AML.
To conclude, we have identified the transcription factor ZFP91 as a novel prominent activator of EVI1 expression in some EVI1-associated AML. Our study suggests that ZFP91 may be the key to unraveling the pathogenesis of EVI1-associated AML and an attractive therapeutic target in EVI1-associated AML.
Disclosures
Kurokawa:Pfizer Japan Inc.: Honoraria; Nippon Kayaku Co., Ltd.: Honoraria; Nippon Shinyaku Co., Ltd.: Honoraria; Kyowa Kirin Co., Ltd.: Honoraria, Research Funding; MOCHIDA PHARMACEUTICAL CO.,LTD.: Honoraria; Janssen Pharmaceutical K.K.: Honoraria; Shionogi & Co., Ltd.: Research Funding; Bristol-Myers Squibb K.K.: Honoraria; Teijin: Research Funding; Chugai Pharmaceutical Co., Ltd.: Honoraria, Research Funding; Sanofi K.K.: Honoraria; Sumitomo Pharma Food & Chemical Co., Ltd.: Honoraria, Research Funding; DAIICHI SANKYO COMPANY, LIMITED: Honoraria, Research Funding; Takeda Pharmaceutical Company Limited.: Honoraria, Research Funding; ONO PHARMACEUTICAL CO., LTD.: Honoraria; Otsuka Pharmaceutical Co., Ltd.: Honoraria, Research Funding; Eisai Co., Ltd.: Honoraria; AbbVie GK: Honoraria, Research Funding; Amgen Inc.: Honoraria; AstraZeneca K.K.: Research Funding; Astellas Pharma Inc.: Honoraria; Asahi Kasei Pharma Corporation: Honoraria, Research Funding; MSD K.K.: Honoraria.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal